For Authors
Getting Started
Interested in submitting to this journal? We recommend that you review the About the Journal page for the journal's aim & scope, as well as the Author Guidelines. Authors need to register with the journal prior to submitting or, if already registered, can simply log in and begin the five-step process.
Submission of Articles
Articles should be submitted online via Online Journal System (OJS) Platform: https://efi.org.in/journal/index.php/JEFI/about/submissions.
Before submitting your article, please read and carefully follow the Instructions for Authors outlined below. All articles should be submitted in English only. If English is not your first language, you are encouraged to seek a professional editing service before submission. If the quality of English is not considered adequate, the manuscript will be returned without review. Authors are given the opportunity to re-submit a revised version that has been edited and improved. Manuscripts may only be actively under consideration by one journal at any given time.

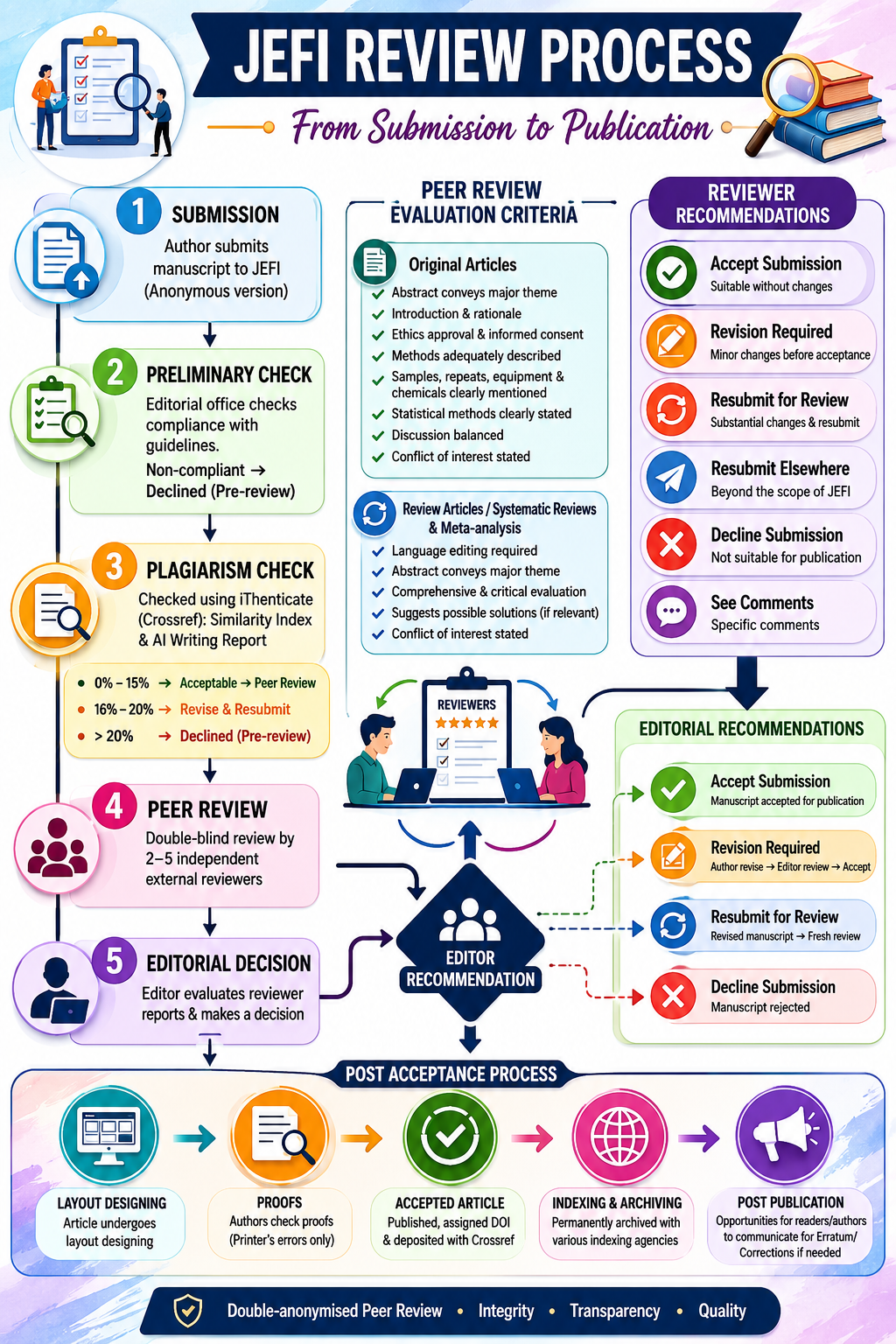
Above image is AI generated summary for JEFI peer review, Editorial Board have reviewed the content.
Review Process
All submissions to JEFI are subject to double-anonymised peer review which requires authors to submit an anonymous version of their manuscript file (to be uploaded as the Article File including abstract). All submitted manuscripts will undergo a three-step review process: i) preliminary check, ii) plagiarism check and iii) peer review.
Preliminary Check
All submitted manuscripts will be reviewed by the journal’s editorial office for compliance with guidelines for preparation of articles. Articles that do not comply with the guidelines will be Declined (pre-review) & sent back to the authors.
Plagiarism Check
Articles that are in compliance with the guidelines will be subjected to plagiarism check. We work with Crossref to use iThenticate to detect plagiarism. iThenticate gives a 'similarity index' & 'AI writing report', which is the word-by-word copying of materials from previously published literature. For articles with similarity index, we follow the COPE Guidelines to make a decision. JEFI Editorial Board follows following similarity index range to make decision.
- 0% – 15% Similarity: Acceptable & will be send for peer-review.
- 16% – 20% Similarity: Will be sent back to the authors for revision & resubmit.
- Over 20% Similarity: Will be Declined (pre-review) & sent back to the authors.
The JEFI has adopted the following policies, as specified by the International Committee of Medical Journal Editors (ICMJE) Section V (Jan 2026), on the use of artificial intelligence (AI) in preparation of material to be submitted for publication in the Journal.
- Authors must disclose at submission of the manuscript whether AI-assisted technologies (such as large language models, chatbots, or image creators) were used to produce the submitted work. If so, both the cover letter and the submitted work should include a description of the technologies used and what was produced.
- Because the authors of a manuscript are responsible for the accuracy, integrity, and originality of the work, chatbots or other AI-assisted technologies cannot be listed as authors.
- Authors should carefully review and edit all materials produced through the use of AI, to prevent the submission of authoritative-sounding output that is incorrect, incomplete, or biased.
- Authors should be able to assert that there is no plagiarism of text or images in materials produced by AI. Authors must ensure that all quoted material is properly attributed, including full citations.
- Citation of AI-generated material as a primary source is not acceptable.
Peer Review
JEFI employs a double-blind peer review process, in which the author identities are concealed from the reviewers, and vice versa, throughout the review process. The entire editorial workflow is performed using OJS. Once a manuscript is submitted, the manuscript is assigned to an Editor most appropriate to handle it based on the subject of the manuscript and the availability of the Editors. If the Editor determines that the manuscript is not of sufficient quality to go through the normal review process or if the subject of the manuscript is not appropriate to the journal scope, the Editor rejects the manuscript with no further processing.
If the Editor determines that the submitted manuscript is of sufficient quality and falls within the scope of the journal, he/she assigns the manuscript to a minimum of 2 and a maximum of 5 external reviewers for peer-review.
The reviewers submit their reports on the manuscripts along with their recommendation of one of the following actions to the Editor:
Accept Submission / Revision Required / Resubmit for Review / Resubmit Elsewhere / Decline Submission / See Comments
Selection of reviewers
JEFI Editorial Team will find appropriate experts to review the manuscripts. Authors may suggest potential reviewers; however, there is no guarantee that these reviewers will be invited to review. Potential reviewers will be personally contacted by the editorial staff to seek expression of interest to review the paper. The email will contain the details of the authors and the abstract. Also, the reviewers will be asked to disclose conflict of interest. If the reviewers agree to review, and if there is no potential conflict of interest, they will receive the official invitation from the journal containing the link to access the manuscript.
Reviewer responsibilities
All reviewers are requested to adhere to a set of basic principles and standards during the peer review process; these are based on the COPE Ethical Guidelines for Peer Reviewers. The reviewers will remain anonymous. Reviewers are expected to keep the manuscript confidential, provide an unbiased scientific opinion of the manuscript and declare any conflict of interest. Also, reviewers are expected to submit their comments in a positive, impartial attitude within the indicated time frame.
Criteria for peer review of articles based on the guidelines provided by Council of Science Editors)
Reviewer invitation
Initial Considerations before accepting a review invitation, please ensure:
Availability and Timeliness: JEFI requests article review response within one week and completion of review within two weeks. If you require a longer period or are unable to meet the deadline, please contact the editor.
Confidentiality: Reviewers are obligated to maintain confidentiality throughout the review process. Do not share, discuss, or disclose any information about the manuscript with others without prior permission from the editorial office.
Misconduct: If reviewer suspect any form of misconduct, such as plagiarism or data fabrication, it must be informed to the editor with specific details.
Use of AI: All evaluations must be conducted personally by the reviewer. Reviewer should not use AI tools for revieing whole manuscript or part of manuscript, as human expertise and critical judgment are essential to maintaining the integrity and quality of the peer review process
Conflict of Interest: Inform the editor if any conflicts exist.
Manuscript File Commenting: Reviewers are requested to provide specific comments or suggested edits directly within the blinded manuscript file using comments or track changes, in addition to their structured review form.
Reviewer Manuscript Evaluation Criteria
Reviewers should assess the manuscript objectively and constructively, focusing on its suitability for JEFI.
Overall Quality and Suitability
Originality: Does the manuscript present original data or analysis not previously published, and does it offer a unique perspective or address a gap in current knowledge?
Innovation: Does the manuscript present novel ideas or findings? Does it introduce innovative methods, perspectives, or interpretations?
Technical Merit: Are the scientific methods and techniques appropriate and robust?
Applicability: Does the study offer practical implications or applications in real-world public health settings?
Presentation and English: Is the manuscript clearly written and grammatically correct?
Relevance to Public Health: Is the content aligned with the scope and mission of Community Medicine and Public Health?
References: Are they latest, appropriate, and correctly formatted?
Section-wise Evaluation
Title: Specific, aligned with the article’s content
Abstract: describes study’s purpose, hypothesis, key methods, findings, conclusions and MeSH keywords within the word limit of 250.
Introduction: Clearly outlines the rationale and public health relevance of the study, supported by appropriate references. Should highlight gaps in existing knowledge and end with clearly stated objectives.
Methods: Detailed methodology including study plan, materials and methods used. Explain sample size justification with sampling technique used. Elaborate ethical considerations.
Results: See if correct statistical analysis used with appropriate interpretation. Are findings given in the tables and graphs matches with the content given?
Discussion and Recommendations: Interprets the key findings in context with existing literature. Highlights the strengths, limitations, and relevance of the study.
Recommendations: Balanced, evidence-based and based on the study findings.
References: Vancouver style, citations numbers in superscripted square brackets
Tables & Figures: Clear, well-labelled, with APA style.
Reporting Guidelines: Encourage authors to use relevant reporting guidelines (e.g., STROBE for observational studies, CONSORT for trials, PRISMA for reviews). The Equator initiative (https://www.equator-network.org) provides accessible tools.
Criteria for assessing original articles
Does the abstract convey the major theme of the paper?
Does the manuscript require language editing?
Does the introduction describe the rationale for the study in the context of the available literature?
Where relevant, have appropriate ethics approval and informed consent been obtained?
Are the methods adequately described?
Is the number of samples, number of repeats, equipment and chemicals used clearly mentioned?
Are statistical methods clearly stated?
Is the discussion well-balanced in light of the available literature and the research findings?
Is conflict of interest stated?
Criteria for assessing review articles, systematic reviews & meta-analysis.
Does the manuscript require language editing?
Does the abstract convey the major theme of the article?
Does the article comprehensively and critically evaluate an existing problem in the context of the available literature?
If relevant does the article suggest a possible solution to the problem?
Is conflict of interest stated?
Reviewer Report Format
Providing Specific Feedback: Justify the recommendations given by the reviewers. When making specific comments, consider numbering your points and referring to page and line numbers in the manuscript.
Constructive Tone: Provide positive feedback first. Use respectful language. Frame suggestions as recommendations, distinguishing between essential revisions (major compulsory) and desirable ones (minor essential or discretionary).
Reviewer Ethical and Confidential Conduct
Maintain strict confidentiality
Avoid using manuscript content
Report ethical concerns to the editor
Reviewer Recommendations
Accept Submission: Suitable without changes
Revision Required: Minor changes before acceptance
Resubmit for Review: Substantial changes & resubmit
Resubmit Elsewhere: Article is beyond the scope of JEFI.
Decline Submission: Not suitable for publication
See Comments: Specific comments
Editorial Decision
The editor gratefully receives a reviewer’s recommendations, but since the editorial decisions are usually based on evaluations derived from several sources, a reviewer should not expect the editor to honour his or her every recommendation. When all reviewers have submitted their reports, the Editor can make one of the following editorial recommendations and the corresponding author will be notified of the decision.
Editorial Recommendations
If the Editor recommends “Accept Submission” the manuscript is accepted for publication.
If the Editor recommends “Revision Required” the authors are notified to prepare and submit a final copy of their manuscript with the required changes suggested by the reviewers. Only the Editor reviews the revised manuscript after the changes have been made by the authors. Once the Editor is satisfied with the final manuscript, the manuscript can be accepted.
If the Editor recommends “Resubmit for Review” the authors are notified to prepare and submit a revised copy of their manuscript with the required changes suggested by the reviewers. The revised manuscript after the changes have been made by the authors wiil again go under a fresh peer review.
If the Editor recommends "Decline Submission", the manuscript is immediately rejected. Also, if two of the reviewers recommend Decline Submission, the manuscript is immediately rejected.
The editorial workflow gives the Editors the authority in rejecting any manuscript because of inappropriateness of its subject, lack of quality, or incorrectness of its results. The journal does not make editorial decisions on the basis of the interest of a study or its likely impact. Studies must be scientifically valid; for research articles this includes a scientifically sound research question, the use of suitable methods and analysis, and following community-agreed standards relevant to the research field. Review process is to ensure a high-quality, fair, and unbiased double blinded peer-review process of every manuscript submitted to the journal, since any manuscript must be recommended by one or more (usually two or more) external reviewers along with the Editor in charge of the manuscript in order for it to be accepted for publication in the journal.
Submitting a revised manuscript
The revised version of the manuscript should be uploaded online only via author dashboard. When submitting a revised manuscript, contributors are requested to include, the ‘referees’ remarks along with point-to-point clarification at the beginning in the revised file itself. In addition, they are expected to mark the changes as underlined or colored text in the article.
Post Acceptance
Each accepted article will undergo layout designing. No change in the names of the authors (by way of additions and deletions) is permissible after acceptance of article.
Proofs
Authors of accepted articles are supplied of printer's proofs online on their Author dashboard and will also receive email alerts when proofs are available. Corrections on the page proofs should be restricted to printer's errors only and no substantial additions/deletions should be made.
Accepted Article
Each accepted article will be published & assigned a DOI, deposited with Crossref and permanently archived with various abstracting and indexing agency.
Moreover, even the finally published articles’ readers and authors have ample opportunities to review the publications and correspondingly communicate to JEFI in case if the published materials correspondingly warrant any publication of erratum/corrections in future JEFI issues.
In-house submissions
In-house submissions that contain the work of any editorial board member are not allowed to be reviewed by that editorial board member, and an independent editor makes all decisions regarding this manuscript. In addition, these manuscripts are reviewed by two external reviewers. This is also disclosed in the published manuscript under the section of Conflict of Interest.
Article Processing Charges (APC)
JEFI is an open access journal which does not levy any Article Processing Charge (APC). There are no submission or page charges, and no colour charges. To support an Open Access System and in order to cover offset expenses for journal management systems, pre-publication production, publication and archiving, the Epidemiology Foundation of India (EFI) covers the publication cost of accepted manuscripts.
Preparation of Manuscript
American spellings should be used. Authors are requested to adhere to the word limits. Authors must mention the word count on the main article file. Articles exceeding the word limit for a particular category of the manuscript would not be processed further. Uniform Requirements for Manuscripts (URM) submitted to Biomedical Journals should be consulted before submission of the manuscript (http://www.icmje.org). All articles should mention how the human and animal ethical aspect of the study was addressed. Whether informed consent was taken or not? Identifying details should be omitted if they are not essential. When reporting experiments on human subjects, authors should indicate whether the procedures followed were in accordance with the Helsinki Declaration of 1975, as revised in 2000. (http://www.wma.net).
Formatting: The manuscript should be double-spaced, with a 2.5 cm margin, 12-point Calibri, and justified. The pages of the manuscript should be numbered on the bottom right corner. Each of the following sections should begin on a separate page. There should be uniform continuous double line spacing maintained throughout the manuscript along with Line numbering.
Language and Writing Style: The manuscript should be written in American English. The author should write the full term for each abbreviation at its first use in the title, abstract, keywords, and text separately unless the abbreviation is a standard unit of measure. The use of acronyms and abbreviations must be kept to a minimum. When used, they are defined at first mention, followed by the acronym or abbreviation in parentheses. If a brand name is cited, supply the manufacturer’s name and address (city, state, and country). Manuscripts will be altered to meet the style guidelines of the JEFI. The authors are requested to check the manuscript for spelling, grammar, and punctuation errors before submission. Headings and titles must be in sentence case and not in Capitals.
Online Submission Preparation Checklist
All manuscripts submitted for publication to the JEFI should include the following:
- Title page file: Covering letter and Title page. (Template)
- Article file: Manuscript with Abstract & Key words. (Template)
- Tables and Figures: should be submitted in supporting material in Image section (word document format). Figures (excluding graphs) should be submitted in Image section (jpeg format with minimum 300 dpi).
- Ethical Clearance Certificate file: A scanned image (pdf)
- Contributor Form File: Signed by all authors to be uploaded. (Template)
Please follow these guidelines for a smooth online digital contributor form submission process: [Coming Soon]
The submitting author must ensure that all co-authors, with their correct email addresses [mandatory], are added in the 'Authors and Institutions' section during the third step of online submission.
After submission, all co-authors will receive an email with a link to provide their digital copyright consent. Please ensure all co-authors complete this process within seven days of submission.
The submitting author can track the copyright status of their co-authors in the 'Manuscript Information' page by clicking the manuscript ID on their dashboard.
Do not upload scanned copies of signed copyright forms during submission.
Manuscript review will proceed only after all co-authors have submitted their digital copyright consent.
In the event that the article is accepted for publication, authors will transfer, assign or otherwise convey all copyright ownership, including any and all rights incidental thereto, exclusively to the Journal.
Title Page File
The covering letter should explain why the paper should be published in the JEFI, rather than a specialty journal. One of the authors should be identified as the corresponding author of the paper, who would be responsible for the contents of the paper as for communication with the Editorial office. Author should declare that the article was not published or under consideration, in part or whole, simultaneously in any other journal or proceedings.
The Title page should contain the title of the manuscript, a short title (not more than 40 letters) to be used as the running title, the Word count of the abstract and main text, the number of references, figures, and the table should also be mentioned in the title page.
Title page should include (i) Name(s) of author(s); (ii) Highest degree; (iii) Name(s) of the Department(s); (iv) Designations (academic position) of authors in the Department; (v) Complete postal addresses, mobile number and e-mail id of all authors; (vi) name of corresponding author with all above mentioned details.
Title page also should include: (i) Type of manuscript: original article/ review/ correspondence/ perspective/ view point/ clinical image/ letter to editor; (ii) Title; (iii) Short title; (iv) Number of Tables; (v) Number of Figures; (vi) Source of financial support in the form of grants; (vii) Registration number in case of Clinical Trials;
Specific author's contribution should be given at the end in the Title page.
Article File
Title
The title of the article should be short, continuous (broken or hyphenated titles are not acceptable), and yet sufficiently descriptive and informative so as to be useful in indexing and information retrieval.
A short running title not exceeding 6-7 words must also be provided.
Abstract
This should be a structured condensation of the work not exceeding 200 words for Reviews Article /Systematic Reviews /Original Article /Letter to Editor. It should be structured under the following headings: Background, Materials & Methods, Results, Conclusions, and 5-8 keywords to index the subject matter of the article arranged alphabetically. Please do not make any other heading.
Text: It must be concise and should follow the IMRAD format: Background, Materials and Methods, Result, Discussion & Conclusion. The matter must be written in a manner, which is easy to understand, and should be restricted to the topic being presented. Each Table and Figure should be on a separate page and should be given at the end of the manuscript after the references. Please do not insert tables & Figure etc. within the text or attach/upload them separately.
Introduction
A brief introduction stating the scope of the paper precisely should be given. Review of the literature should be restricted to reasons for undertaking the present study and provide only the most essential background. The objective of the study should be written clearly with adequate justification at the end of this section.
Material & Methods
The nomenclature, the source of material and equipment used, within the manufacturer's details in parenthesis, should be clearly mentioned. The procedures adopted should be explicitly stated to enable other workers to reproduce the results, if necessary. New methods may be described in sufficient detail indicating their limitations. Established methods can be just mentioned with authentic references and significant deviations, if any given, with reasons for adopting them. While reporting experiments on human subjects and animals, it should be clearly mentioned that the procedures followed are in accordance with the ethical standards laid down by the national bodies or organizations of the particular country. For example, for research carried out in India on human subjects, the ICMR's Ethical guidelines for biomedical and health research on human participants (2017) should be adhered to Click Here for view. Similarly, for experiments on laboratory animals the ICMR's guidelines: Use of animals in scientific research (May 2006)/INSA's guidelines for care and use of animals in scientific research (2000) or guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA: https://ccsea.gov.in/Auth/index.aspx) should be followed. Adequate information should be provided on the care and use of laboratory animals, source of animals, strain, age, sex, housing and nutrition, etc. Whenever needed, appropriate certification should be provided at the time of submission of the manuscripts. The drugs and chemicals used should be precisely identified, including generic name(s), dosage(s), and route(s) of administration.
Study design: Selection of the observational or experimental participants (patients or laboratory animals, including controls, whether randomly or consecutively) and basis of sample size calculation should be mentioned clearly, including eligibility and exclusion criteria and a description of the source population.
Reporting Guidelines for specific study designs:
|
SN |
Type of Study |
Source |
|
1. |
Randomized controlled trials (RCTs) |
CONSORT- http://www.consort-statement.org |
|
2. |
Systematic reviews & meta-analysis |
PRISMA guidelines- http://www.prisma-statement.org |
|
3. |
Observational studies in epidemiology |
STROBE - http://www.strobe-statement.org/ |
|
4. |
Meta-analysis of observational studies in epidemiology |
|
|
5. |
Studies on diagnostic accuracy |
STARD - http://www.stard-statement.org |
* For any other type of study contributors may consult ICMJE website (www.icmje.org)
Clinical Trials: All clinical trials should be registered in a Primary Clinical Trial Registry and the Registration number should be given under Material & Methods.
Articles presenting with results of randomized clinical trials should provide information on all major study elements, including the protocol, assignment of interventions (methods of randomization, concealment of allocation to treatment groups), and the method of masking (blinding), based on the CONSORT Statement (http://www.consort-statement.org/). It should be clearly stated that study protocol was approved by the institutional/local ethics committee and written consent obtained from the participants.
The statistical analysis done and statistical significance of the findings when appropriate, should be mentioned. Unless absolutely necessary for a clear understanding of the article, detailed description of statistical treatment may be avoided. Articles based heavily on statistical considerations, however, need to give details particularly when new or uncommon methods are employed. For standard and routine statistical methods employed, authors need to give only authentic references.
Results
Only such data as are essential for understanding the discussion and main conclusions emerging from the study should be included. The data should be arranged in unified and coherent sequence so that the report develops clearly and logically. Data presented in Tables and Figures should not be repeated in the text. Only important observations need to be emphasized or summarized. The same data should not be presented both in tabular and graphic forms. Interpretation of the data should be taken up only under the Discussion and not under Results.
Discussion
The discussion should deal with the interpretation of results without repeating information already presented under Results. It should relate new findings to the known ones and include logical deductions. It should also mention any weaknesses/limitations/lacunae of the study.
Conclusion
The conclusions can be linked with the goals of the study but unqualified statements and conclusions not completely supported by the data should be avoided. Claiming of priority on work that is ongoing should also be avoided. All hypotheses should, if warranted, clearly be identified as such; recommendations may be included as part of the Discussion, only when considered absolutely necessary and relevant. This section should preferably end with a concluding remark.
Limitation of the study
Explains the weaknesses or constraints of the research that may affect the interpretation or generalization of the findings.
Authors Contribution
How each author have contributed in this study as per ICMJE guidelines for authorship contribution.
Financial support & Sponsorship
A statement should be made for funding support and /or sponsorship received from national or international funding agencies.
Conflict of interest
A brief statement on the source of funding and conflict of interest should be included. It should be included on a separate page immediately following the title page.
Acknowledgment
These should be placed as the last element of the text before references. Written permissions of persons/agencies acknowledged should be provided.
AI Declarations
During the preparation of this work, the author (s) used (mention the name of tool/service used) in order to (mention the reason for using AI tool). After using this tool/service, the author (s) reviewed and edited the content as needed and take (s) full responsibility for the content of the publication.
References
In citing other work only reference consulted in the original should be included. If it is against citation by others, this should be so stated. Signed permission is required for use of data from persons cited in personal communication. ANSI standard style adapted by the National Library of Medicine (NLM) should be followed. (http://www.nlm.nih.gov/bsd/uniform_requirements.html)
References should be numbered and listed consecutively in the order in which they are first cited in the text and should be identified in the text, tables and, legends by Arabic numerals as superscripts in square brackets (Vancouver). The full list of references at the end of the paper should include; the names and initials of all authors up to six (if more than 6, only the first 6 are given followed by et al.); the title of the paper, the journal title abbreviation according to the style of Index Medicus (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=journals), year of publication; volume number; first and last page numbers. Reference of books should give the names and initials of the authors, book title, place of publication, publisher, and year; those with multiple authors should also include the chapter title, first and last page numbers, and names and initials of editors. For citing website references, give the complete URL of the website, followed by the date of accession of the website. Quote such references as – the author's name, the title of the article, the website address, and the date of accession.
Example: …… with no evidence of intratubular testicular neoplasia.(1)
Unpublished data or articles submitted for publication are not to be cited in the reference list. They are cited within parenthesis in the text. (Example: Aaron J, unpublished data). Papers presented at meetings are not cited in the reference list. They are cited within parenthesis in the text. (Example: Aaron J et al., presented at the 2009 annual meeting of the Society).
References follow the ICMJE guidelines. The author’s surname is followed by the author’s initials in capitals without spaces or full stops. All references show page numbers in the format (121-26). Refer to the List of Journals Indexed in Index Medicus for abbreviations of journal names, or access the list from here.
Sample references are given below:
Articles in Journals
1. Olson MC, Posniak HV, Fisher SG, ME Flisak, CG Salomon, RC Flanigan, et al. Directed and random biopsies of the prostate: indications based on combined results of transrectal sonography and prostate-specific antigen density determinations. Am J Roentgenol 1994;163:1407–11.
List the first six contributors followed by et al in all references.
Issue with supplement
Payne DK, Sullivan MD, Massie MJ. Women’s psychological reactions to breast cancer. Semin Oncol 1996;23(1, Suppl 2):89-97.
Volume with supplement
Shen HM, Zhang QF. Risk assessment of nickel carcinogenicity and occupational lung cancer. Environ Health Perspect 1994;102 Suppl 1:275-82.
Books and Other Monographs
Personal Author(s):
Ringsven MK, Bond D. Gerontology and leadership skills for nurses. 2nd ed. Albany (NY): Delmar Publishers; 1996.
Editor(s), compiler(s) as author:
Norman IJ, Redfern SJ, editors. Mental health care for elderly people. New York: Churchill Livingstone; 1996.
Chapter in A Book
Turgut AT, Dogra VS. Prostate carcinoma: Evaluation using transrectal sonography. In: Hayat MA, ed. Methods of cancer diagnosis, therapy and prognosis. 1st ed. New York, NA: Elsevier; 2008. p. 499-520.
Electronic Sources as Reference
Refer APA’s Quick guide on reference for electronic references.
Monograph on the Internet format:
Foley KM, Gelband H, editors. Improving palliative care for cancer [monograph on the Internet]. Washington: National Academy Press; 2001 [cited 2002 Jul 9].
Refer Homepage/Web site and Part of a homepage/Web site formats in the hyperlink provided
- Journals: Mehta MN, Mehta NJ. Serum lipids and ABO Blood group in cord blood of neonates. Indian J Pediatr. 1984; 51:39-43.
- Book: Smith GDL. Chronic ear disease. Edinburgh: Churchill Livingstone; 1980.
- Chapter in the Book: Malhotra KC. Medicogenetics problems of Indian tribes. In: Verma IC, editor. Medical genetics in India. vol. 2. Pondicherry: Auroma Entrprises; 1978. p. 51-55.
- Papers accepted but not yet published should be included in the references followed by 'in press'. Those in preparation, personal communications, and unpublished observations should be referred to as such in the text only.
Tables and Figures
The count of tables/ figures are restricted to Editorial (1), Review article/Systematic Reviews/Meta-Analysis/Original articles(5) and Letter to Editor(1).
Tables: These must be self-explanatory and must not duplicate information in the text. Each table must have a title and should be numbered with Arabic numerals. Each table should be typed in double space, on a separate page and included at the end of the manuscript after the references. No internal horizontal or vertical lines should be used. All tables should be cited in the text. Tables should be prepared in APA standard format. Tables should be numbered consecutively with Arabic numerals (1,2,3…etc). They should bear a brief title and column headings should also be short. Units of measurement should be abbreviated and placed below the headings. Statistical measurement variations such as SD and SE should be identified. Inclusion of structural formula in Tables should be avoided. Abbreviations used to be given in the footnote.
Illustration / Figures: These should be of the highest quality, submit glossy black and white photographs. Graphs should be drawn by the artist or prepared using standard computer software. Number all illustrations with Arabic numerals (1,2,3….). Figures should be submitted in JPEG or TIFF format (size not more than 1 MB and minimum 300 dpi) with appropriate Title and explanation of symbols in the legends for illustrations. Within a multi-panel figure, different parts should be labelled as A, B, C,...etc. on top left corner. Photomicrographs should have internal scale markers regarding details of magnification to facilitate reduction in size in final print. Symbols, arrows and letters used in the photomicrographs should be legible and in contrast with the background. Graphs in JPEG/TIFF format can be uploaded as Figures.
All published material should be acknowledged and copyright material should be submitted along with the written permission of the copyright holder.
Legends: A descriptive legend must accompany each illustration and must define all abbreviations used therein.
Click here to download the powerpoint presentation on common reference styles and using the reference checking facility on the manuscript submission site.
Abbreviation: The abbreviations should be used in the text, tables and illustrations without a full stop. Authors should define new abbreviations when used first in the text. Standard abbreviations to be used in JEFI
|
Molar (mole/litre) |
M* |
counts per minute |
cpm |
|
milli molar (m mole/litre) |
mM |
Curie |
Ci |
|
Micromolar (mole/litre) |
μM |
rad |
rad |
|
mole (quantity of substance) |
mol |
Roentgen |
R |
|
normal |
N |
gravity |
g |
|
metre |
m |
ortho |
o |
|
centimetre |
cm |
meta |
m |
|
square centimetre |
cm2 |
para |
p |
|
millimetre |
mm |
intramuscular |
im |
|
micrometre |
μm |
intraperitoneal |
ip |
|
nanometre |
nm |
intravenous |
iv |
|
picometre |
pm |
subcutaneous |
sc |
|
mg/ 100 ml |
mg/dl |
oral |
po |
|
Angstrom |
Å |
lethal dose-50 |
LD50 |
|
litre |
1 |
Ampere |
A |
|
millilitre |
ml |
milli Ampere |
mA |
|
microlitre |
μl |
Watt |
W |
|
gram |
g |
anti meridiem (before noon) |
am |
|
milligram |
mg |
post meridiem (after noon) |
pm |
|
kilogram |
kg |
volume |
vol |
|
hour(s) |
h |
volume ratio |
vol/vol |
|
minute(s) |
min |
(volume per volume) |
|
|
second(s) |
sec |
weight |
wt |
|
week(s) |
wk |
weight per volume |
wt/vol |
|
year(s) |
yr |
weight ratio |
wt/wt |
|
Probability |
|
|
|
|
(statistical significance) |
P |
(weight per weight) |
|
*Should not be used as an abbreviation for mole.
Ethical Clearance Certificate File
All studies conducted on patients/ volunteers/ Human biological material/animals should submit a scanned copy of Ethical Clearance Certificate.
Contributor Form File
It is necessary that all the authors give an undertaking (in the format specified by the journal) indicating their consent to be co-authors in the sequence indicated on the title page. Each author should give his or her names as well as the address and appointment at the time the work was done, additionally current address for correspondence including telephone and email address. A senior author may sign the Undertaking by Authors for a junior author who has left the institution and whose whereabouts are not known and take responsibility.
A paper with corporate (collective) authorship must specify the key persons responsible for the article; others contributing to the work should be recognized separately.
Author(s) will be asked to sign a transfer of copyright agreement, which recognizes the common interest that both journal and author(s) have in the protection of copyright. It will also allow us to tackle copyright infringements ourselves without having to go back to authors each time.
Types of Manuscripts
|
Article type |
Guidelines |
Pages* |
Word limit |
Figure / Table |
References |
Abstract |
|
Editorial |
Editorial should take a proactive approach to making the situation better by using constructive criticism and giving solutions. |
1-3 |
500- 1500 |
1-2 |
10 |
Unstructured 100 words |
|
Review Article |
A review is a comprehensive overview of the literature that synthesizes the current research, presents the key findings, and highlights gaps in the existing literature. Narrative reviews are an important tool for researchers to identify research trends and to inform future research directions. |
6-10 |
3500 - 5000 |
Up to 5 |
50 |
Structured 200 words (Background, Aims & Objectives, Material & Methods, Results, Conclusion) |
|
Systematic Reviews |
A systematic review is a rigorous and comprehensive analysis of existing research on a specific topic. It involves identifying, selecting, and appraising multiple studies that meet predetermined inclusion criteria. The purpose of a systematic review is to summarise the available evidence and provide an unbiased and reliable assessment of the current state of knowledge. |
6-10 |
3500 - 5000 |
Up to 5 |
50 |
Structured 200 words (Background, Aims & Objectives, Material & Methods, Results, Conclusion) |
|
Meta-Analysis |
A meta-analysis is a statistical method that combines results from multiple independent studies to produce a single, more precise estimate of an effect or relationship.
|
6-10 |
3500 - 5000 |
Up to 5 |
50 |
Structured 200 words (Background, Aims & Objectives, Material & Methods, Results, Conclusion) |
|
Original Article |
Original research articles report original data. This article type includes all research reviews that systematically synthesise evidence. Research submissions should have a clear, justified research question. IMRaD style (Introduction, Methods, Results and Discussion) |
5-6 |
3500 |
Up to 5 |
35-50 |
Structured 200 words (Background, Aims & Objectives, Material & Methods, Results, Conclusion) |
|
Letter to Editor |
|
2 |
1000 |
1 |
10 |
No |
*Number stated refers to the maximum number of typeset pages.
Clinical Trials Registration
The International Committee of Medical Journal Editors (ICMJE) and, therefore, JEFI require investigators to register trials in acceptable clinical trial registries before the onset of patient enrollment. Manuscripts describing primary results of nonregistered trials will be turned away prior to peer review.
Clinical Trials
The ICMJE requires that “any research project that prospectively assigns human subjects to intervention and comparison groups to study the cause-and-effect relationship between a medical intervention and a health outcome” must be registered before the start of patient enrollment. To be acceptable, a registry must be owned by a non-for-profit entity, be publicly accessible, and contain the 20 fields required by the World Health Organization (WHO). View a list of acceptable registries on the WHO website.
Each manuscript will be checked on submission to determine whether the study needed registration, and, if registered, whether the registration is complete and meaningful. No manuscript will enter the editorial process until it has passed this screen.
Pilot Trials
Pilot (phase 1) trials that began on or after July 1, 2008 must be registered before first patient enrollment.
Acceptable Registries
WHO maintains a list of acceptable registries. In addition, ICMJE will accept registration in any of the primary registries that participate in the WHO International Clinical Trials Registry Platform (ICTRP).
Authorship Criteria
JEFI follows ICMJE recommended 4 authorship criteria:
- Substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work; AND
- Drafting the work or revising it critically for important intellectual content; AND
- Final approval of the version to be published; AND
- Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
In addition to being accountable for the parts of the work he or she has done, an author should be able to identify which co-authors are responsible for specific other parts of the work. In addition, authors should have confidence in the integrity of the contributions of their co-authors.
For Authorship: All those designated as authors should meet all four criteria for authorship, and all who meet the four criteria should be identified as authors. Those who do not meet all four criteria should be acknowledged. These authorship criteria are intended to reserve the status of authorship for those who deserve credit and can take responsibility for the work. The criteria are not intended for use as a means to disqualify colleagues from authorship who otherwise meet authorship criteria by denying them the opportunity to meet criterion #s 2 or 3. Therefore, all individuals who meet the first criterion should have the opportunity to participate in the review, drafting, and final approval of the manuscript.
Non-Author Contributors: Contributors who meet fewer than all 4 of the above criteria for authorship should not be listed as authors, but they should be acknowledged. Examples of activities that alone (without other contributions) do not qualify a contributor for authorship are acquisition of funding; general supervision of a research group or general administrative support; and writing assistance, technical editing, language editing, and proofreading. Those whose contributions do not justify authorship may be acknowledged individually or together as a group under a single heading (e.g. "Clinical Investigators" or "Participating Investigators"), and their contributions should be specified (e.g., "served as scientific advisors," "critically reviewed the study proposal," "collected data," "provided and cared for study patients", "participated in writing or technical editing of the manuscript").
Contribution Details: Contributors should provide a description of contributions made by each of them towards the manuscript. Description should be divided in following categories, as applicable: concept, design, definition of intellectual content, literature search, clinical studies, experimental studies, data acquisition, data analysis, statistical analysis, manuscript preparation, manuscript editing and manuscript review. One or more author should take responsibility for the integrity of the work as a whole from inception to published article and should be designated as 'guarantor'.
Archiving | Abstracting & Indexing
Archiving | Abstracting | Indexing of JEFI is currently with Google Scholar's Citations l Crossref l ISSN l Geneva Foundation for Medical Education and Research l Scilit l Index Copernicus l Open Alex (Supported by Arcadia, The Navigation Fund, and The Wellcome Trust) l Elsevier Solutions | Scite | SciSpace | ivySCI | ePUBLISH INDIA | OpenAIRE | Keepers | Road (Directory of Open Access Scholary Resources)
Open Alex: H-index - 5; i10-index: 2, 02 Year mean citedness: 0.339 |
Archiving | Abstracting | Indexing of JEFI in processes with J-Gate | Web of Science (WoS) | Directory of Open Access (DOAJ) | PubMed | EMBASE | Scopus | JSTOR | Portico | Artstor | ResearchGate | Indian Citation Index (ICI) | EBSCO Host | CLOCKSS | CABI | International Journal Indexing (IJI) | BASE | CORE Indexing
Ahead of Print Policy
The editors of JEFI are pleased to offer electronic publication of accepted titles prior to issue publication. Articles appearing in “Ahead of Print” section have been peer-reviewed and to achieve faster and greater dissemination of knowledge and information, the journal publishes articles online as ‘Ahead of Print’ immediately on acceptance. Articles appearing here may contain statements, opinions, and information that have errors in facts, figures, or interpretation.
Journal reprints
Journal reprints provide scientific evidence that establishes epidemiological ground breaking research. JEFI provide reprint services including articles, supplements, entire issue reprints, and conference proceedings on demand.
Archiving Policy
JEFI archives its content with LOCKSS.
The LOCKSS program offers decentralized and distributed preservation, seamless perpetual access, and preservation of the authentic original version of the content.
The PKP PN ensures that journals that are not part of any other digital preservation service (such as CLOCKSS or Portico) can be preserved for long-term access. For additional details about the PKP PN, see the high-level overview (an early discussion document). All URLs are OAI-qualified
Privacy Statement
The names and email addresses entered in this journal site will be used exclusively for the stated purposes of this journal and will not be made available for any other purpose or to any other party.
Open Access Statement
This is an open access journal which means that all content is freely available without charge to the user or his/her institution. Users are allowed to read, download, copy, distribute, print, search, or link to the full texts of the articles, or use them for any other lawful purpose, without asking prior permission from the publisher or the author. This is in accordance with the Budapest Open Access Initiative BOAI definition of open access.
Copyright
All of the content published in the JEFI is protected under the Indian and International copyright law, defined by Creative Commons and International Committee of Medical Journal Editors (ICMJE). The author of an article retains the academic copyright of the content and can self-archive the article. The journal retains the commercial rights of the published content and publisher executes the commercial rights on behalf of the journal.
The journal also grants to all readers and users a free, irrevocable, global, perpetual right of access to, and a license to copy, use, distribute and display the content publicly and to make and distribute derivative works in any digital medium for any reasonable and non-commercial purpose, subject to proper attribution of authorship and ownership of the copyrights under the Creative Commons Attribution-Noncommercial-NoDerivatives 4.0 International Public License.
- Share — copy and redistribute the material in any medium or format for any purpose, even commercially.
- The licensor cannot revoke these freedoms as long as you follow the license terms.
Under the following terms:
- Attribution: You must give appropriate credit, provide a link to the license, and indicate if changes were made. You may do so in any reasonable manner, but not in any way that suggests the licensor endorses you or your use.
- Non-commercial: You may not use the material for commercial purposes.
- No Derivatives: If you remix, transform, or build upon the material, you may not distribute the modified material.
- No additional restrictions: You may not apply legal terms or technological measures that legally restrict others from doing anything the license permits.
- Notices: You do not have to comply with the license for elements of the material in the public domain or where your use is permitted by an applicable exception or limitation. No warranties are given. The license may not give you all of the permissions necessary for your intended use. For example, other rights such as publicity, privacy, or moral rights may limit how you use the material.
Compliance with Funder-Mandated Open Access Policies
An author whose work is funded by an organization that mandates the use of the Creative Commons Attribution-Non-Commercial-Share Alike 4.0 License is able to meet that requirement through the available open-access license for approved funders.
Protection of Patients' Right to Privacy
Identifying information should not be published in written descriptions, photographs, sonograms, CT scans, etc., and pedigrees unless the information is essential for scientific purposes and the patient (or parent or guardian, wherever applicable) gives informed consent for publication. Authors should remove patients' names from figures unless they have obtained informed consent from the patients. The journal abides by ICMJE guidelines:
- Authors, not the journals nor the publisher, need to obtain the patient consent form before the publication and have the form properly archived. The consent forms are not to be uploaded with the cover letter or sent through email to editorial or publisher offices.
- If the manuscript contains patient images that preclude anonymity or a description that has an obvious indication of the identity of the patient, a statement about obtaining informed patient consent should be indicated in the manuscript.
Ethical Guidelines
JEFI adheres to the highest standards concerning its editorial policies on publication ethics, scientific misconduct, consent and peer review criteria. The journal follows guidance produced by bodies that include the Committee on Publication Ethics (COPE), the World Association of Medical Editors (WAME), the Council of Science Editors and the International Committee of Medical Journal Editors (ICMJE).
Authors are required to submit a statement that their study obtained ethics approval from IRB (or a statement that it was not required and why) and that participants gave written informed consent. Editors will consider whether the work is morally acceptable as determined by the World Medical Association’s Declaration of Helsinki. In addition to this, in line with General Medical Council guidelines, an article that contains personal medical information about an identifiable living individual requires patients’ explicit consent before we will publish it.
The key documents on research ethics (such as The Belmont Report, the Declaration of Helsinki, the GCP Guidelines and the CIOMS Guidelines) stressed the importance of including vulnerable subjects (such as pregnant women, children below 18 years, geriatric population, prisoners, foetuses, and individuals with consent capacity impairment) in research, with the due precautionary measures.
To make the best decision on how to deal with a manuscript, we need to know about any competing interests authors may have; this includes any commercial, financial or non-financial associations that may be relevant to the submitted article. Authors must download and complete a copy of the ICMJE Conflict of Interest disclosure form. In addition to this we ensure that all advertising and sponsorship associated with the journal does not influence editorial decisions, is immediately distinguishable from editorial content.
JEFI will not consider publication of papers reporting work funded wholly or partly by the tobacco industry. This also excludes work where the authors have personal financial ties with the tobacco industry. This applies to all content types.
These guidelines are intended to provide a framework for ethical behaviour and decision-making. We expect all contributors, and partners to adhere to these guidelines and uphold the highest standards of ethical conduct.
Plagiarism
JEFI is extremely strict in matters of Plagiarism. The Journal will not accept plagiarized articles for publication. If the journal finds a submitted plagiarized article, then it may ban all the authors responsible for the article in publishing with it. So, all the authors/researchers are requested to check their respective articles for plagiarism before submitting them for publishing.
JEFI has also joined Crossref - iThenticate Similarity Check in the battle against plagiarism and to guarantee high ethical standards for all of the published papers. iThenticate Turnitin is a powerful tool for identifying plagiarised material, allowing our editors to protect the integrity of the journal and the authors' copyright.
We shall carry out detection on approved articles. A fair citation was generally considered to be one that did not exceed 200 words of overlap between the paper and the literature. When substantial amounts of text overlap are discovered (e.g., >5% overlap or >10% similarity), we immediately check to see if the article is linked to publishing misconduct. We may immediately enforce the aforementioned sanctions if publication misbehaviour with the material is discovered.
All submitted manuscripts in JEFI will be vigorously checked for plagiarism using the iTHENTICATE plagiarism detection software before forwarding the manuscripts for the peer-review process.
All allegations of plagiarism are investigated in accordance with COPE guidelines detailed below:
- COPE guidelines on suspected plagiarism in a submitted manuscript
- COPE guidelines on suspected plagiarism in a published paper
- ICMJE Recommendations
- UGC guidelines for plagiarism
In the case of suspected plagiarism in a published article:
- The person who advised us of the situation is informed about the process to be followed.
- The articles are compared to check the degree of copying.
- All Editors of the JEFI are informed and asked for their comments.
- The corresponding author of the article in question is contacted with documentary evidence of the case of plagiarism and is asked for a response.
- If the authors are found guilty of plagiarism
- The editor of the journal in which the original plagiarised article was published and the authors of the plagiarised article are informed
- The JEFI publishes an official retraction of the paper
- The online version of the JEFI article is withdrawn from the website, and
- The JEFI will not publish any article by any of the authors concerned for a period of five years.
Ethics and Misconduct
JEFI has a zero-tolerance policy for publications linked to publication misconduct in order to protect the standards of academic integrity and respect for other people's intellectual property rights. Plagiarism, fabrication, falsification, inappropriate authorship, duplicate submission/multiple submissions, overlapping publishing, and salami publication are all examples of publication misconduct. We take all possible cases of misconduct seriously. If an Editor, author or reader has concerns that a submitted article contains something that might be considered to constitute misconduct in research, publication or professional behaviour they should forward their concerns to the journal. The editorial board will deal with allegations appropriately following ICMJE and COPE guidelines.
According to COPE Committee criteria for publishing misconduct, we have created JEFI definitions and policies, which are as follows:
- Plagiarism: The unjustified use of another person's words, ideas, data, statistics, research techniques, or other work without giving due credit, as well as the excessive citation of another person's published work.
- Fabrication: Without conducting the pertinent study, fabrication is the technique of inventing data or outcomes.
- Falsification: Falsification is the deliberate alteration of facts or results to lead to false conclusions.
- Inappropriate authorship: The author's contributions are not taken into account for determining authorship.
- Duplicate submission/multiple submissions: Refer to the act of submitting the same manuscript or multiple manuscripts with minor differences (e.g., differences only in title, keywords, abstract, author order, author affiliations, or a small amount of text) to two or more journals at the same time, or submitting to another journal within an agreed-upon or stipulated period.
- Overlapping publication: Publishing a paper that considerably overlaps with one that has already been published is referred to as an overlapping publication.
- Salami publication: Salami publication is the process of dividing data from a large study into smaller portions that could have been reported in a single paper and publishing them in two or more articles that all address the same population, techniques, and question.
- Cross-referencing references with the text
- What If There Was Misconduct?
- Immediate Rejection the manuscript or revoke the published article.
- Not accept submissions submitted within two years by the same research team.
- Informing the institution of the corresponding author.
Policy for handling errata, retractions, withdrawals, and expressions of concern
JEFI abide by COPE Retraction Guidelines. These policies are intended to provide a framework for addressing issues that may arise with published content. We will follow these policies with care and transparency, and communicate clearly with all stakeholders throughout the process.
Complaint’s procedure
- The complaint may be submitted via email to the journal editor at [email protected]
- The investigation involves reviewing all correspondence relating to the case in question and, if necessary, obtaining further written responses to queries from the parties involved.
- The purpose of the investigation is to establish that correct procedures have been followed, that decisions have been reached based on academic criteria, that personal prejudice or bias of some kind has not influenced the outcome, and that appropriate sanctions have been applied where relevant.
- Complainants may choose to take their complaint to the Committee on Publication Ethics (COPE).
It is important to establish clear ethical guidelines to ensure that all content is produced and distributed in a responsible and ethical manner.
- Accuracy and fact-checking: We strive to publish accurate and truthful content, and we take all necessary steps to fact-check information before publication. We correct any errors promptly and transparently.
- Editorial independence: We maintain editorial independence and do not allow advertisers, sponsors, or any external parties to influence our content in any way.
- Transparency: We disclose any conflicts of interest and relationships with external parties that may influence our content, and we provide clear attribution for sources and contributors.
- Respect for diversity and inclusivity: We respect and celebrate diversity in all its forms, and we strive to represent diverse perspectives and voices in our content.
- Protection of personal information: We respect individuals’ right to privacy and we protect personal information in accordance with applicable laws and regulations.
- Copyright and intellectual property: We respect the intellectual property rights of others and do not infringe on any copyrights or trademarks in our content.
- Non-discrimination: We do not publish content that discriminates against any individual or group based on race, ethnicity, gender, sexual orientation, religion, or any other protected characteristic.
- Responsibility for content: We take responsibility for all content published under our name, and we promptly address any complaints or concerns related to our content.
Errata: An erratum is a correction of a factual error that does not alter the overall conclusions of a published work. When an error is identified, we will promptly correct the error and publish an erratum that clearly identifies the correction and its implications, such changes are intimated to respective indexing agencies and DOI records are updated.
Retractions: A retraction is a notice that a published work is no longer valid or reliable. Retractions may be necessary when a work contains serious errors, plagiarism, or other forms of misconduct. The publisher and the editorial team of the journal will investigate any concerns raised about published content and, if necessary, issue a retraction that clearly explains the reasons for the retraction, such changes are intimated to respective indexing agencies and DOI records are updated.
Process for issuing a retraction statement: Where the decision is taken to retract and the article to be retracted is the Version of Record (i.e. it has been published as Online First or within an issue of a journal), National Academy of Medical Sciences recommends issuing a retraction statement which should be published separately and should be linked to the article being retracted. A "retracted" watermark should also be added to the article; however, the article as first published should be retained online to maintain the scientific record. Issuing a retraction statement will mean the following:
- The retraction will appear on a numbered page in a prominent section of the journal.
- The retraction will be listed in the contents page and the title of the original article will be included in its heading.
- The text of the retraction should explain why the article is being retracted; and
- The statement of retraction and the original article must be clearly linked in the electronic database so that the retraction will always be apparent to anyone who comes across the original article.
Withdrawals: A withdrawal is a notice that a published work is being removed from circulation because of ethical concerns or legal issues. Withdrawals may be necessary when a work contains fraudulent or unethical research or when legal issues arise. We will investigate any concerns raised about published content and, if necessary, issue a withdrawal that clearly explains the reasons for the withdrawal.
Any manuscript before or post acceptance can be withdrawn with a clear reason and the editorial team of the respective journals take appropriate action. National Academy of Medical Sciences. as a publisher do not interfere in any decision making during these process. The DOI (Digital Object Identifier) is assigned only at the time of online publication.
Expressions of Concern: An expression of concern is a notice that a published work is being reviewed for potential problems, but no decision has been made about whether to retract or correct the work. Expressions of concern may be necessary when there are concerns about the validity or reliability of a published work. The publisher and the editorial team will investigate any concerns raised about published content and, if necessary, issue an expression of concern that clearly explains the reasons for the concern and the steps being taken to address the issue.
Conflicts of Interest
All authors of must disclose any and all conflicts of interest they may have with publication of the manuscript or an institution or product that is mentioned in the manuscript and/or is important to the outcome of the study presented. Authors should also disclose conflict of interest with products that compete with those mentioned in their manuscript.
To ensure unbiased evaluations, we require all reviewers to disclose any potential conflicts of interest that could influence their judgment during the review process.
Artificial Intelligence (Generative AI) Policy
The JEFI has adopted the following policies, as specified by the International Committee of Medical Journal Editors (ICMJE), on the use of artificial intelligence (AI) in preparation of material to be submitted for publication in the Journal.
- Authors must disclose at submission of the manuscript whether AI-assisted technologies (such as large language models, chatbots, or image creators) were used to produce the submitted work. If so, both the cover letter and the submitted work should include a description of the technologies used and what was produced.
- Because the authors of a manuscript are responsible for the accuracy, integrity, and originality of the work, chatbots or other AI-assisted technologies cannot be listed as authors.
- Authors should carefully review and edit all materials produced through the use of AI, to prevent the submission of authoritative-sounding output that is incorrect, incomplete, or biased.
- Authors should be able to assert that there is no plagiarism of text or images in materials produced by AI. Authors must ensure that all quoted material is properly attributed, including full citations.
- Citation of AI-generated material as a primary source is not acceptable.
For further guidance, please refer to the Ethical Best Practices in Scholarly Publishing for policy on generative AI use for authors and International Committee of Medical Journal Editors (ICMJE) Section V (Jan 2026), on the use of artificial intelligence (AI) in preparation of material to be submitted for publication in the Journal.

